
CFDNA ISOLATION (PLASMA, URINE, CELL CULTURE MEDIA)
 CBI offers a new system for the isolation of circulating nucleic acids (DNA and RNA) from liquid biopsies, cells and tissues. Our DNA isolation kit is based on a new DNA Binding Substance that captures circulating DNA from biofluids (serum, plasma, urine, amniotic and cerebrospinal fluids) without addition of salts and/or chaotropic agents. No Proteinase K digestion is required. Standard volume of liquid biopsy for DNA isolation is 2-5 ml. Scale up to 50 ml of liquid can easily be done.
CBI offers a new system for the isolation of circulating nucleic acids (DNA and RNA) from liquid biopsies, cells and tissues. Our DNA isolation kit is based on a new DNA Binding Substance that captures circulating DNA from biofluids (serum, plasma, urine, amniotic and cerebrospinal fluids) without addition of salts and/or chaotropic agents. No Proteinase K digestion is required. Standard volume of liquid biopsy for DNA isolation is 2-5 ml. Scale up to 50 ml of liquid can easily be done.
We have developed a new method of cell-free DNA (cfDNA) isolation from plasma, serum, and urine samples based on a dual-functional proprietary substance (SubX) that binds DNA under physiological conditions (e.g., directly from biological fluids) where it is absorbed to a solid phase Binding Matrix, e.g. magnetic beads. Current commercial cfDNA isolation kits employ a traditional silica-chaotropic salt approach and often suffer from lot-to-lot surface differences of silica that result in low extraction efficiency of small DNA fragments. Our approach eliminates this problem.
Our DNA isolation procedure includes the following steps:
• DNA binding: simply add SubX and Binding Matrix to your bioliquid sample and rotate for a few minutes;
• Washing: brief vortex-spins;
• Elution: DNA is easily eluted in a small volume (30-50ul) of buffer.
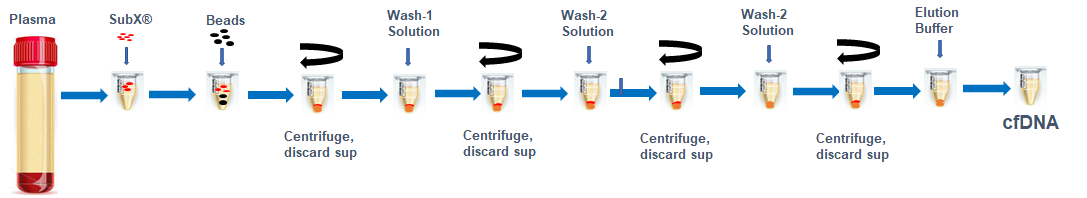
Figure 1. DNA isolation steps. All procedures after mixing plasma or urine with SubXTM are done in one tube. Each incubation takes place at room temperature with occasional vortexing. cfDNA is eluted from beads at 65ºC.
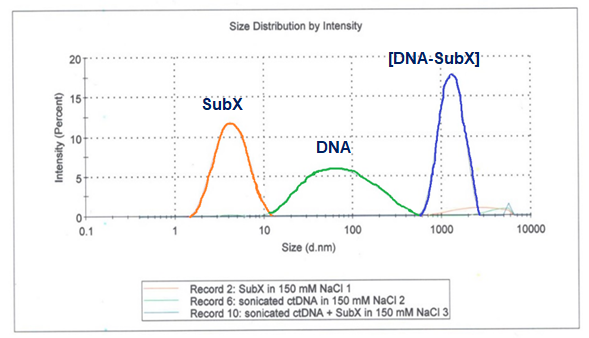
Figure 2. Interaction of SubXTM with sonicated DNA analyzed using Zetasizer Nano ZS.
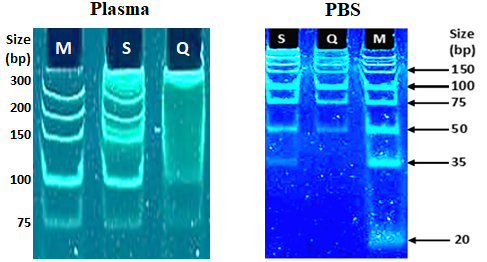
Figure 3. Short DNA fragments recovery from plasma and PBS. M- Ultra Low DNA Ladder (ThermoFisher); S- spike DNA isolated with SubXTM cfDNA kit; Q- DNA isolated with kit from vendor Q.
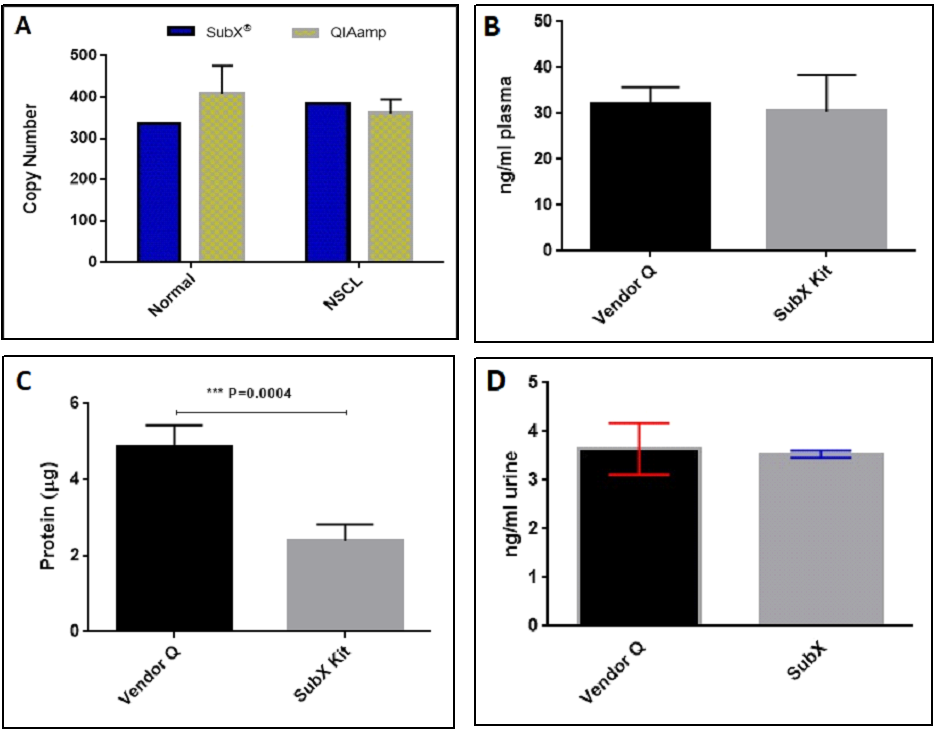
Figure 4. DNA yield from 0.2 ml plasma of normal donors and NSCL patients estimated by qPCR for single copy gene 36B4 (A). cfDNA yield from 1 ml pooled plasma determined by Qubit HS DNA Assay (B). Protein content in cfDNA Isolated from pooled plasma (C). cfDNA isolated from 4 ml urine samples. DNA was quantified using Qubit HS DNA Assay (D).
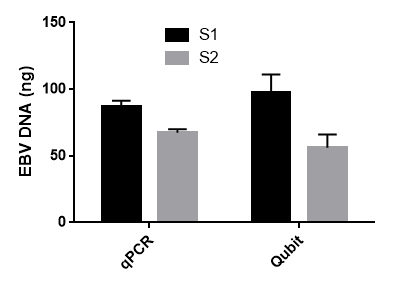
Figure 5. Eppstein Barr Virus DNA isolation from B95-8 cell line culture medium. DNA was isolated from 3x1ml aliquots of cell culture S1 and S2. DNA yield was estimated by qPCR with EBV-specific primers and by Qubit HS DNA Assay.
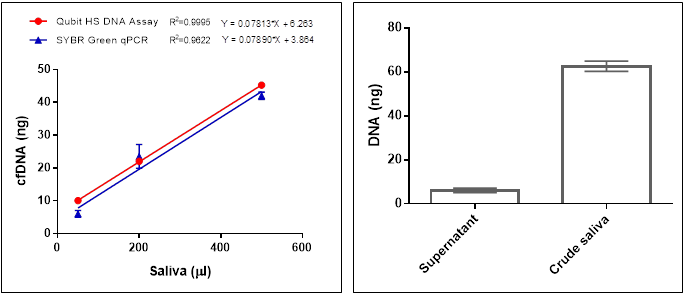
Figure 6. DNA yield from saliva was determined by fluorescent Qubit HS DNA Assay and real time SYBR Green qPCR with primers for human single copy gene 36B4 (left). Comparison of the DNA yield from crude saliva and from supernatant after centrifugation at 16,000xg for 1 min (right). DNA was isolated from 50 ml aliquots diluted with 950 ml PBS. DNA was determined by real time SYBR Green qPCR with primers for human single copy gene 36B4.

